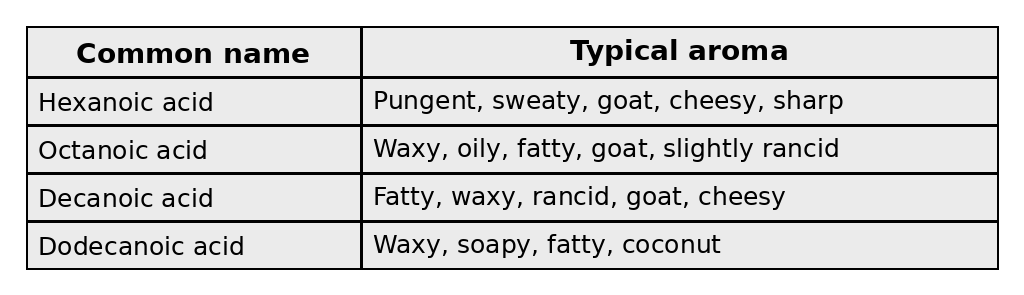Making Funky, High Ester Rum - Part 1
Flavor Chemistry and Yeast Species
Sometimes my procrastination about starting to write subconsciously signals that I have more mental homework to do. So it was, while procrastinating over a story about a new high-ester rum, that I realized I needed to further hone my understanding of some nerdy, wonk fundamentals.
I’ve written quite a few stories here about rum aromas, flavors, and their associated chemistry. Still, in outlining how to approach this new rum, I realized my earlier writings had hand-waved past some important concepts. Back to the proverbial woodshed I went, forcing myself to head-on face dark corners I’d previously ignored. The result is a series of stories, starting with this one, that introduce basic fermentation chemistry for laypeople, then navigate through the techniques rum makers use to create extreme rums. To use a car analogy, how they turn a Toyota Camry into a top-fuel funny car.
What follows below establishes a baseline for how the majority of rum makers ferment their source material. Subsequent posts will explore how creating highly flavored, high-ester rums differs from mainstream rum making.
To start, let’s review a tiny bit of flavor chemistry to better understand what follows.
Flavor Basics
Fundamentally, the smell and taste of an unaged rum is the result of a soup of organic compounds generated during fermentation. This soup’s components are then filtered and concentrated via distillation.
When these organic compounds contact our nasal receptors and tongue, our nose and tongue send signals to our brain, which interprets them as smell and taste.
The primary organic compounds found in distilled spirits span several classes: acids, alcohols, esters, and aldehydes. Each class, e.g., alcohols, has subtypes that vary in molecular structure and complexity. For example, methanol and ethanol are both alcohols, but ethanol has more carbon and hydrogen atoms than methanol. As such, ethanol is “heavier” than methanol.
Here are four of the most molecularly simple alcohols:
Here are four of the most molecularly simple acids:
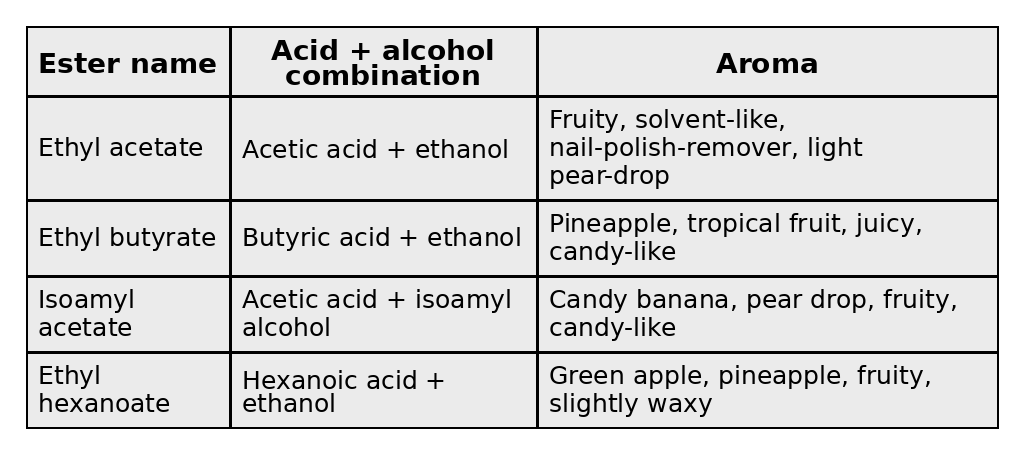
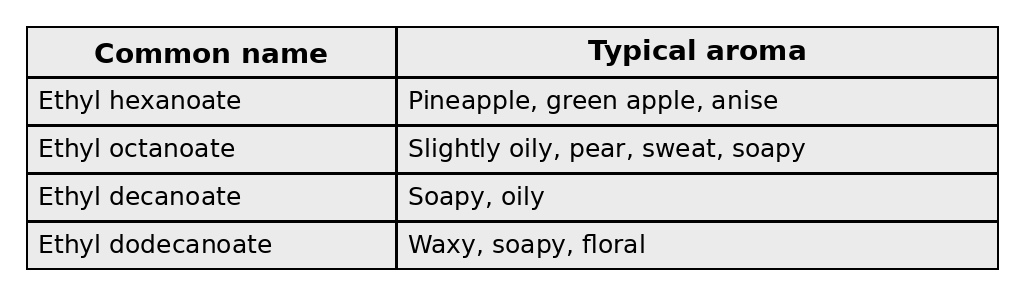
When an alcohol and an acid combine, they form an ester. Here are four typical esters you’d find in everyday, middle-of-the-road rums.
There will be no test on the above, but it is important to grasp that the flavor in distilled spirits derives from a complex stew of different organic compounds, including those above. Each organic compound is like an instrument in an orchestra, making its own contribution to the symphony your nose and tongue experience. (Yes, I’m purposefully omitting flavors induced by aging. That’s another story for another time.)
It’s also important to know that these compounds contribute nearly all of a spirit’s distinctive aroma and flavor character while making up less than 1% of its volume. 99% of a distilled spirit is either ethanol or water, and neither has a particularly strong aroma or flavor. The chemical difference between a bottle of Bacardi Superior and a bottle of Hampden HLCF would fit in a thimble with room to spare.
Now, where do all these organic compounds come from?
The Mighty Yeast Cell
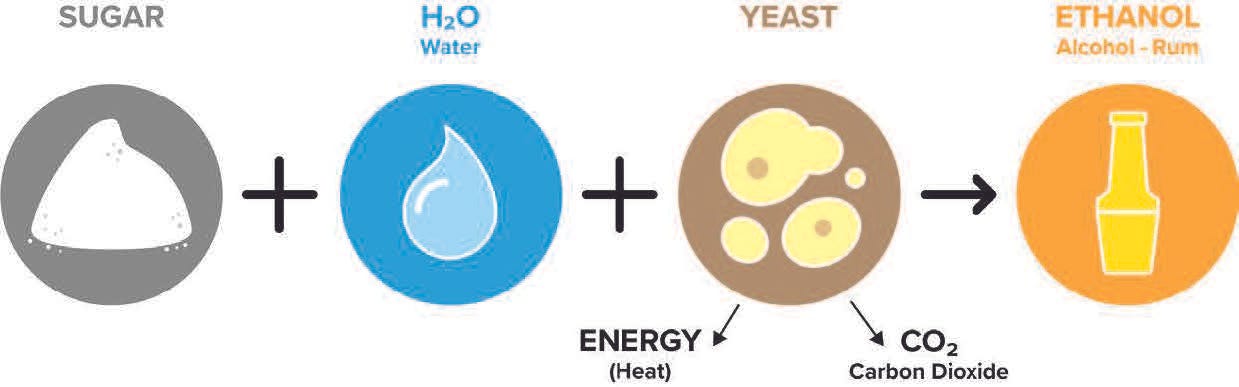
When people first learn about rum, fermentation is usually described like this:
“Molasses and water are mixed, after yeast is added, the result is a fermenting mash. The yeast consumes the sugar and emits alcohol.”
While not incorrect, it dramatically undersells the story; it’s like saying Oppenheimer was a movie about a science project.
Yeast cells are microscopic superfactories squeezed into a single-cell organism. Inside their walls, they feed on sugar and other mash compounds and emit a mix of different flavor compounds along with ethanol. They even knit molecules together within the cell, turning the alcohol and acid molecules they previously made into esters.
Yeast cells also emit heat and carbon dioxide during fermentation. If you’ve ever wondered what makes bread dough rise, it’s carbon dioxide “bubbles” created by the yeast consuming sugars in the dough.
The types and amounts of organic compounds a yeast cell emits are influenced by its food source, i.e., the sugar molecules and other nutrients in the mash. Differences in the chemical composition of cane juice or molasses can cause yeast to emit a different mix of organic compounds. These differences are one aspect of a spirit’s terroir.
The environment where a yeast cell resides also influences the mix of organic compounds it emits. The temperature of the fermenting mash affects what the yeast creates. If the mash gets too warm, the yeast cells can die. (Remember, fermentation creates heat, and yeast can be responsible for its own death unless steps like cooling the mash are taken.)
Likewise, if the mash’s pH drops too low, the yeast may die — some strains are more tolerant of low pH than others. Even the ethanol the yeast emits can kill it if the mash ABV gets too high.
All that said, a bit of controlled stress in a yeast cell’s life can be a good thing, causing it to emit a more desirable mix of flavor compounds. We’ll look at this in a future installment of this series.
A Brief Burst of Biology and Organic Chemistry
Most rum makers use a yeast strain within a species called Saccharomyces cerevisiae. Here, we’ll call it cerevisiae for brevity. Within this species, you’ll find a variety of strains. Some strains are popular for champagne, others for beer, others for bread, and of course, rum.
The primary emission from cerevisiae yeast is ethanol, as you’d expect. As for the other organic compounds it creates, most are on the simpler and lighter end of the molecular spectrum: acids like acetic acid (vinegar), alcohols like propanol and isoamyl alcohol, and esters formed when those particular acids and alcohols combine. The signature esters produced by cerevisiae, including ethyl acetate and isoamyl acetate, give a typical rum its familiar fruity character.
To rephrase this more holistically, cerevisiae yeast produces aromas reminiscent of banana, honey, juicy fruit gum, and apple. Ethyl acetate, the most common ester in rum, smells lightly fruity at low concentrations but turns to solvent or nail polish remover aromas at higher concentrations. Anyone with experience drinking high-ester rums knows that note well.
The Reigning Champion
The vast majority of the rum world runs on a steady stream of S. cerevisiae yeast. Distilleries pick a strain they like and either carefully maintain it in their laboratories or purchase ready-made strains in dry form. Think a packet of Fleischmann’s Dry Yeast, but on a far larger scale! Yeast specialists like AB Biotek and Lallemand specialize in creating bulk and specialized yeast for distilleries around the globe.
With an eye towards consistency from batch to batch, a few rum makers heat their molasses and water before adding yeast. The heat kills any ambient yeast and bacteria, creating a very clean playing field for the cerevisiae yeast to work in.
The Saccharomyces cerevisiae species is the de facto commodity yeast used by most distilleries because it works quickly, is reliable, is well understood, and is readily available. It creates a consistent result, which large-scale rum makers prize.
The Dark Horse
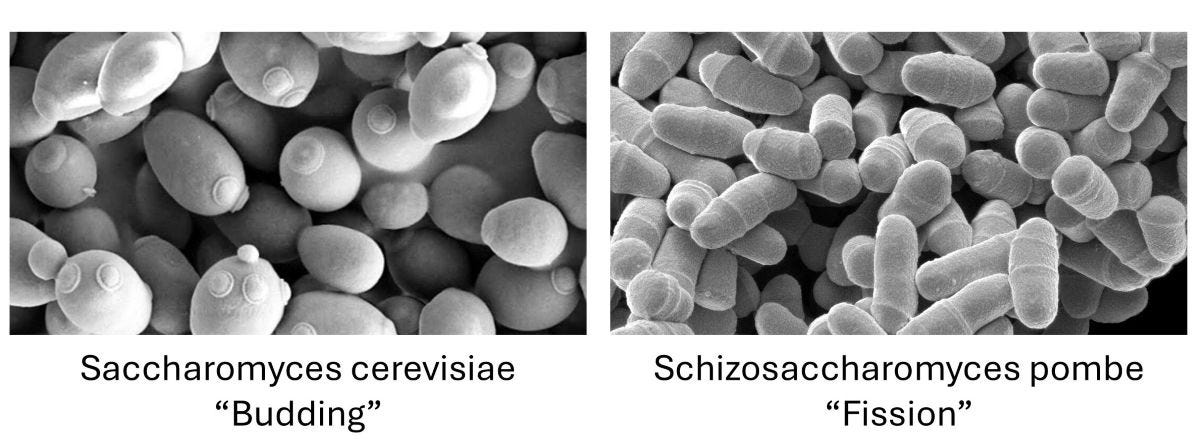
If Saccharomyces cerevisiae strains are the global pop stars of yeast, the punk underground equivalent is Schizosaccharomyces pombe. We’ll call it pombe here for brevity. Taxonomically speaking, pombe is a different species from cerevisiae. A good analogy from the plant kingdom is oak tree vs a sunflower. While they share many common attributes, they are noticeably different in other aspects.
Among the differences is that pombe cells look and reproduce differently from cerevisiae cells. Cerevisiae reproduces by growing small “buds” that pop off and grow to full size, while pombe cells cleave in half, which is why they’re called fission yeast.
If both pombe and cerevisiae are given the same mash, and the resulting washes are chemically compared, we’d usually find different acid and alcohol profiles. Pombe-associated fermentations tend to push the resulting rum toward heavier, more complex aromas, including those coming from medium-chain fatty acids such as:
Similarly, pombe favors creating more complex alcohols, including:
If you’ve heard the terms higher alcohols or fusel alcohols, they are alcohols that are molecularly heavier than ethanol and contribute to spirits that are heavier, oily, and sometimes solventy. Pombe creates a greater percentage of higher alcohols (like the above) than cerevisiae.
Naturally, because pombe produces a different mix of acids and alcohols compared to cerevisiae, the ester profile also changes. Pombe fermentations create more of these esters:
In short, the more molecularly complex mix of organic compounds created by pombe creates a rum with intense tropical-fruit aromas like pineapple and overripe banana, along with a heavier, thicker mouthfeel.
Who’s using pombe yeast today? Jamaica’s rum makers are the flagship pombe users, but it pops up in other regions known for making wild, “weird”, and highly flavorful rums, including Martinique’s Le Galion. Beyond those known users, it’s plausible that Grenada’s River Antoine, Haiti’s clairin producers, and a few Mexican distilleries are also using pombe to some degree, though none have officially confirmed it.
As an aside for any distillers out there, AB Biotek recently released the first commercially available dry Pombe yeast targeted at rum makers:
While pombe yeast creates a heavier and more intense flavor profile, it comes with some operational downsides. While cerevisiae typically finishes fermentation in 1-3 days, pombe can take three or more times as long to complete, while yielding less alcohol. Nonetheless, the divine flavor of Jamaican and other rums makes this tradeoff worth it.
Conclusion
Wrapping things up for this initial installment, the above comparison of pombe and cerevisiae yeast assumes similar mashes and environmental conditions. However, pombe has a superpower that cerevisiae mostly lacks: when pombe cells are stressed in the right way, they turn on the flavor production afterburners. Cerevisiae responds to stress as well, but pombe is much more at home in the low-pH, bacteria-laced conditions associated with some heavy-rum fermentations.
That stress, along with another microbial actor, is where things get really weird, wild, and wonderful. Pombe yeast by itself can’t create high-ester flavor bombs like Hampden DOK or Long Pond TECC. Pombe is often the foundation of such rums, but only when paired with techniques you won’t find in regular rum making. The next installment in this series jumps headfirst into this topic.
A hearty thank you to Brett Steigerwaldt for reviewing the above for me prior to publishing.